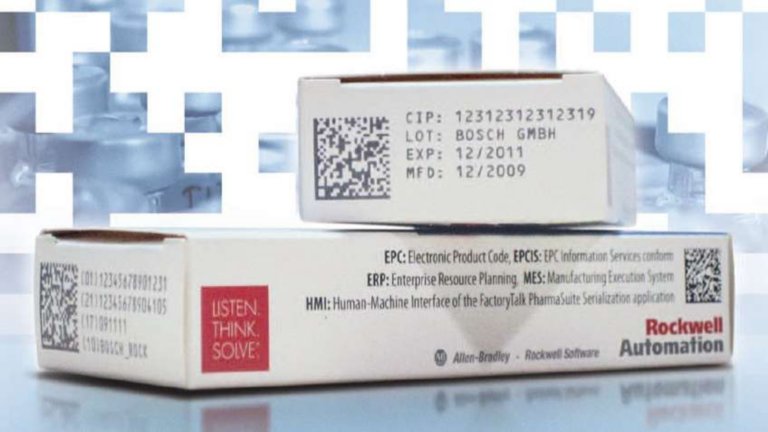
Deal with today's and tomorrow's challenges with a holistic serialization solution that covers automation, MES, ERP and Cloud. Find flexibility and cost savings while positioning yourself to address future regulations, counterfeiting threats and recalls.
Find a Complete Solution for Pharma Serialization
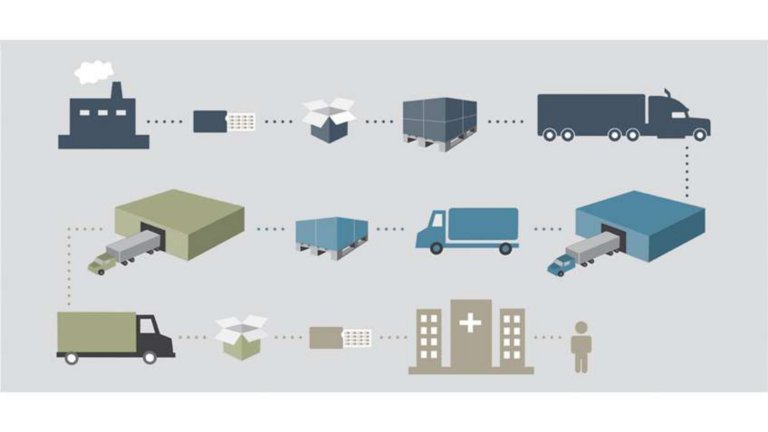
Communication between plant floor and front office has gone from a nice-to-have to a must-have. It is not as difficult as you think to search for a solution that addresses specific business challenges and current regulatory requirements. The ability to have a solution that includes real-time visibility and ISA-95 certification while meeting pharmaceutical serialization requirements is possible.
Serialization Helps Reduce Risk, Improve Profitability
Global pharmaceutical companies lose an estimated $75 billion annually to counterfeit, gray market and stolen product. Regulations will require pharma manufacturers to track and trace products across the supply chain. Our serialization solution takes a holistic approach to product tracking and traceability. We provide a single approach to address regulations, product counterfeiting and recalls.
Scalable, Future-Proof Serialization
Pharma manufacturers need modular, scalable serialization solutions built on an integrated control and information system that can be easily integrated into existing production lines. They need seamless interoperability among machines, devices and business systems. Our serialization solution enables you to comply with current regulations with the flexibility to adapt to future requirements.
Others also viewed
Based on your activity