By Jim Toman, Lead Consultant - Food and Beverage Services and Ian Tooke, Director - Consulting Services, Grantek Systems Integration
The Food Safety Modernization Act (FSMA) will revolutionize food safety in the United States, and will have a ripple effect across the globe. Of the seven rules proposed by the U.S. Food & Drug Administration (FDA) thus far, the majority are hundreds of pages in length, and understanding what's required can be confusing. With additional rules still expected to come, it can be difficult to plan for the future.
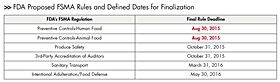
So far, the FDA has released the proposed rules shown in the illustration, with court-defined dates for finalization. While regulatory compliance is a baseline requirement, the ability to concurrently satisfy other business needs helps ease the burden of implementing a change.
Food Safety Evolution
The Hazard Analysis Critical Control Point (HACCP) approach to food safety has been gaining ground over the past several decades, and is required for certain types of food products in the United States (seafood, juice, meat and poultry). FMSA will require the breadth of the food industry to build upon HACCP and formalize a comprehensive system to address food safety. Standards will be instituted for fresh produce, food processing, transportation and imports.
Rather than issuing product-specific requirements, the FDA sets the objective and leaves it up to companies to figure out how to get there. While the FDA doesn't require the use of any specific system, technology, or the use of any degree of automation, it's clear that the FDA wants companies to embrace technology.
Regardless of the approach taken, several overarching “ingredients” are needed for FSMA compliance that also are critical for your brand protection:
-
Rapid Response. Whether it's recognizing that a process is out of control or providing records to the FDA, companies need to be able to respond quickly.
-
Consistency. When considerable effort goes into developing a plan to achieve food safety, the consistent implementation of those policies is critical.
-
Documentation. The foundational role that documentation will play in demonstrating that food safety is under control — even when a regulator or auditor isn't looking — can't be overstated.
-
Analysis. The food industry collects a lot of data. However, bringing the data together to make sense of the bigger picture often is lacking. Trending data allows companies to anticipate looming issues and also can reveal opportunities to optimize systems and efficiency. While FSMA revolutionizes the FDA's food safety authorities, an individual company's path to compliance should be more of an evolution, not a revolution.
Preventive Controls
Preventive controls prevent, eliminate or reduce the known hazard to an acceptable level for safe consumption of food. Under FSMA, the FDA will have a legislative mandate to require comprehensive, science-based preventive controls across the food supply.
Moving forward, some activities that have historically been considered current Good Manufacturing Practices (cGMPs) may be updated and modernized, thus falling under Preventive Controls.
Within the realm of Preventive Controls, process controls are the most familiar. These are the controls that are typically associated with HACCP and are recognized as the critical control points for food safety. With Preventive Controls, the FDA expects that food manufacturers will continue to heavily rely upon process controls. Consistent with what is seen in existing HACCP regulations, the FDA would require that the following approach be followed:
Validation. Whatever process control is selected must work. If a process is being employed, for example, to control for Salmonella, the food producer must be able to show that the process will destroy the pathogen effectively. In this example, this means knowing the expected and worst case levels of the pathogen so that the amount of “kill” can be determined.
Then, the parameters that will accomplish this reduction need to be tested. This should be done using the food products to which the process will be applied, under the most real-to-life conditions possible, including using the actual equipment that will be used during production.
Establishing Parameters. Validation studies will help a company establish the conditions needed for an effective process. Any change, such as a change in formulation or processing equipment, should prompt a re-evaluation of the parameters. Automated recipe management for equipment and process parameters can help food producers meet this need.
Monitoring. Once the parameters are set, they must be monitored. A Historian database can be used to capture these process parameters such as temperature, pressure, time, etc.
Corrective Actions. System failures need to be anticipated, and facilities need to show they have thought through what could go wrong and have a plan to address issues if they occur. When issues do occur, the facility needs to keep records of the event and show they followed the course of action specified in their food safety plan.
Verification. The FDA has proposed a few ways facilities will need to show that their entire food safety system is working as anticipated. Recordkeeping and documentation, and a review of those records as well as a re-evaluation of the overall food safety plan, would be required.
Examples of Preventive Controls
Allergen Control. For facilities that have identified food allergens as hazards that are reasonably likely to occur, implementing allergen controls would be required. This can encompass several different activities, including appropriate sanitation between different allergens as well as runs without allergens, appropriate storage of allergens, material verification, and label control and verification.
Supplier Verification. In some cases, a facility might not have a way to control a hazard, instead relying on a supplier for control. In this instance, the FDA is expected to propose that supplier controls be enacted in such a way that the receiving facility is assured that their supplier is adequately controlling hazards.
Certificates of Analysis (CoAs) by suppliers have little value if a receiving company doesn't know how to review and verify them properly. The preferred approach is to automate the review of COAs. Implementing an automated traceability and genealogy system covering raw materials receipt and linking laboratory analysis of raw materials samples to the specific received lots can create a traceable and searchable record that proves control.
Forensic Traceability. An automated track-and-trace system, with genealogy records including key data elements (KDEs) captured at critical tracking events (CTEs) and further detail of equipment and production routings, can facilitate fast forensic investigation and build trust with the FDA and consumers.
Compliance Isn't Optional
FSMA is coming, and change is just around the corner. Leading companies are continually on the lookout for ways to enhance their operations while maintaining profitability.
Grantek Systems Integration Inc., based in Burlington, Ontario, Canada, is a Rockwell Automation Enterprise Solution Partner, an elite group of professional IT service organizations that have demonstrated design, delivery and sup‐port competency around the enterprise class of Rockwell Automation FactoryTalk® manufacturing information solutions. The firm is a system integrator and solution provider specializing in manufacturing IT solutions, manufacturing automation solutions, industrial safety solutions and manufacturing consulting services.
The Journal From Rockwell Automation and Our ParnterNetwork is published by Putman Media, Inc.